

OTC medicinal products
What are OTC drugs?
In accordance with European legislation, an OTC (over-the-counter) drug is a medicinal product sold without a medical prescription. These drugs are used mainly for purposes of short-term interventional self-medication following consultation with a pharmacist. In certain cases, they can also be used in long-term treatment of diseases – especially chronic ones – under the condition of being supervised by the pharmacist or physician. OTC drugs are readily available and intended for treatment of the most common ailments. They include active ingredients recognised as safe when used according to the guidelines presented in the leaflet in scope of both posology and treatment duration.OTC drug registration
When a marketing authorisation holder wants to market a medicinal product in the non-prescription category, said holder files an application for the appropriate permit to the appropriate national authorities. In Poland, this is the URPL (Urząd Rejestracji Produktów Leczniczych – Office for Registration of Medicinal Products). In Europe, it is EMA (European Medicines Agency). These entities decide on releasing certain medicinal products in OTC status according to the data determining the drug’s safety in scope of strictly limited treatment duration and in ailments, which can be easily self-diagnosed by the patient. OTC switch, which is based on switching the medicinal product category from prescription to OTC, is currently very popular among pharmaceutical companies. This is done according to the proposed range of therapeutic recommendations, the data confirming the safety of the given active ingredient (even in the event of administration of the wrong dose or misunderstood indications), and the patient’s ability to diagnose the disease, which the drug in question is supposed to treat.The OTC market
In 2020, the OTC drug and dietary supplement segment composed approximately 36% of the total pharmaceutical trade in Poland. The growth of the OTC market both in Poland and worldwide was driven by the COVID-19 pandemic, which had particular impact on the segment’s sales in Q1 of 2020, with the most popular being respiratory care products, vitamins and minerals, and painkillers.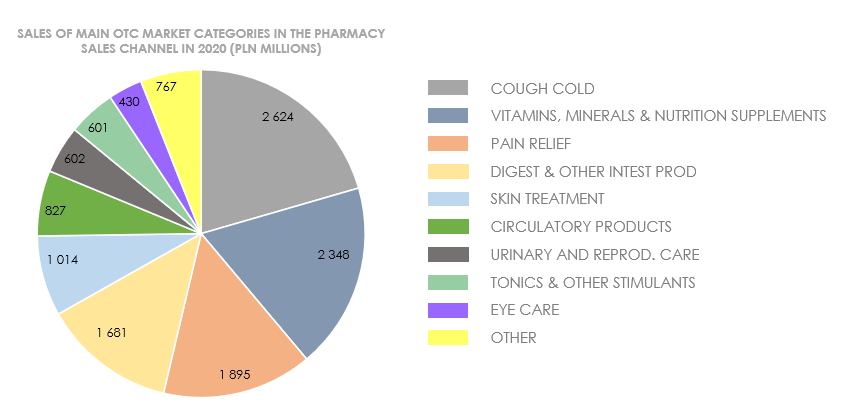
Non-pharmacy marketing
Drugs authorised for non-pharmacy marketing – distribution in supermarket chains, drugstores, small and medium-sized grocery stores, and gas stations – compose a special group of OTC medicinal products. In 2020, their sales composed 6% of the total trade in the OTC segment. OTC marketing involves products, which include active substances and their maximum content listed in the Resolution of the Minister of Health, which in practice means a limited number of dose units (capsules, tablets, or pills). The option of purchasing OTC drugs in non-pharmacy outlets creates a controversy, which has been raising safety concerns in the medical community for years. Besides the extensive availability, which is an unquestionable advantage of this distribution channel[1], its numerous flaws are also subject to discussion: uncontrolled storage conditions (air temperature and humidity) required to ensure proper product quality and unavailability of professional consulting at purchase, which may affect proper product use. In light of the above, the medical community recommends purchasing medicinal products from pharmacies, which guarantee professionalism and top standards.References
[1] Response of the undersecretary of the Ministry of Health – under authorisation of the minister – to interpellation No. 7980 concerning non-pharmacy drug trade.Author
R&D Department, Curtis Health Caps
tel.: +48 61 625 27 00
fax: +48 61 625 27 01
office@chc.com.pl
bd@chc.com.pl
purchasing@chc.com.pl
rd@chc.com.pl
BDO: 000102401
Wysogotowo
ul. Batorowska 52
62-081 Przeźmierowo
POLAND
District Court for Poznań - Nowe Miasto and Wilda in Poznań, the 8th Commercial Division of the National Court
KRS 0000871229
NIP 781-00-41-371
Share capital: 36 100 000PLN
© Curtis Health Caps. All rights reserved.


