

Safety of vitamin D supplementation. Is an overdose possible?
Vitamin D metabolism
Vitamin D obtained after sun exposure and taken with food or dietary supplements is biologically inactive and must undergo activation in two successive enzymatic hydroxylation reactions occurring in the liver and kidney (Fig. 1). Endogenously, vitamin D (cholecalciferol) is photosynthesised in the skin. The 7-dehydrocholesterol (provitamin D) present in the skin is converted to the precalciferol (PreD) form in the epidermis after exposure to ultraviolet B radiation (UVB 290-315 nm) and then to vitamin D (Fig. 1). Vitamin D production in the skin is a result of the extent and quality of UVB radiation reaching the dermis, as well as the availability of 7-dehydrocholesterol and the properties of the skin. Dietary vitamin D is absorbed in the small intestine along with other dietary fats [3]. In the liver, precalciferol is rapidly hydroxylated by 25-hydroxylase to inactive 25-hydroxycholecalciferol (25(OH)D, calcidiol) [4], which, after binding to the protein ( Vitamin D-Binding Protein; DBP), is transported with the blood to the kidneys, where it is hydroxylated by 1,25-hydroxylase to the active form 1,25- dihydroxycholecalciferol (1,25(OH)2D, calcitriol). The average time for 25(OH)D to be present in plasma is about 3 weeks and this is what makes serum 25(OH)D levels an indicator of vitamin D status in the body.Regulation of vitamin D metabolism
The level of the active form of vitamin D (1,25(OH)2D) is very closely regulated by blood calcium and phosphate levels via parathyroid hormone (PTH) and fibroblast growth factor 23 (FGF-23) [5]. Serum levels of vitamin D and 25(OH)D depend on the season and vitamin D intake [5], whereas serum levels of the active metabolite 1,25(OH)2D are invariant (16-65 pg/mL) and independent of sunlight exposure or supplementation. With adequate levels of 25(OH)D in the body, the kidneys play a key role in metabolising 25(OH)D to the biologically active metabolite, 1,25(OH)2D. In the case of hypocalcaemia (low blood calcium levels), there is an increased secretion of PTH and an increase in the activity and rate of renal hydroxylation and formation of the active vitamin D metabolite (1,25(OH)2D), which then increases the reabsorption of calcium from the tubules raising its concentration in the blood (Fig.1).In turn, high levels of 1,25(OH)2D also affect the renal metabolism of 25(OH)D (calcidiol), decreasing the activity of 25-hydrolases and increasing the metabolism of calcidiol to 24,25(OH)2D (the inactive metabolite). This leads to the degradation of both 25(OH)D and 1,25(OH)2D to water-soluble inactive metabolites and their excretion from the body [6]. This, in turn, leads to a reduction in the levels of active vitamin D metabolites in the body and normalisation of blood calcium levels.
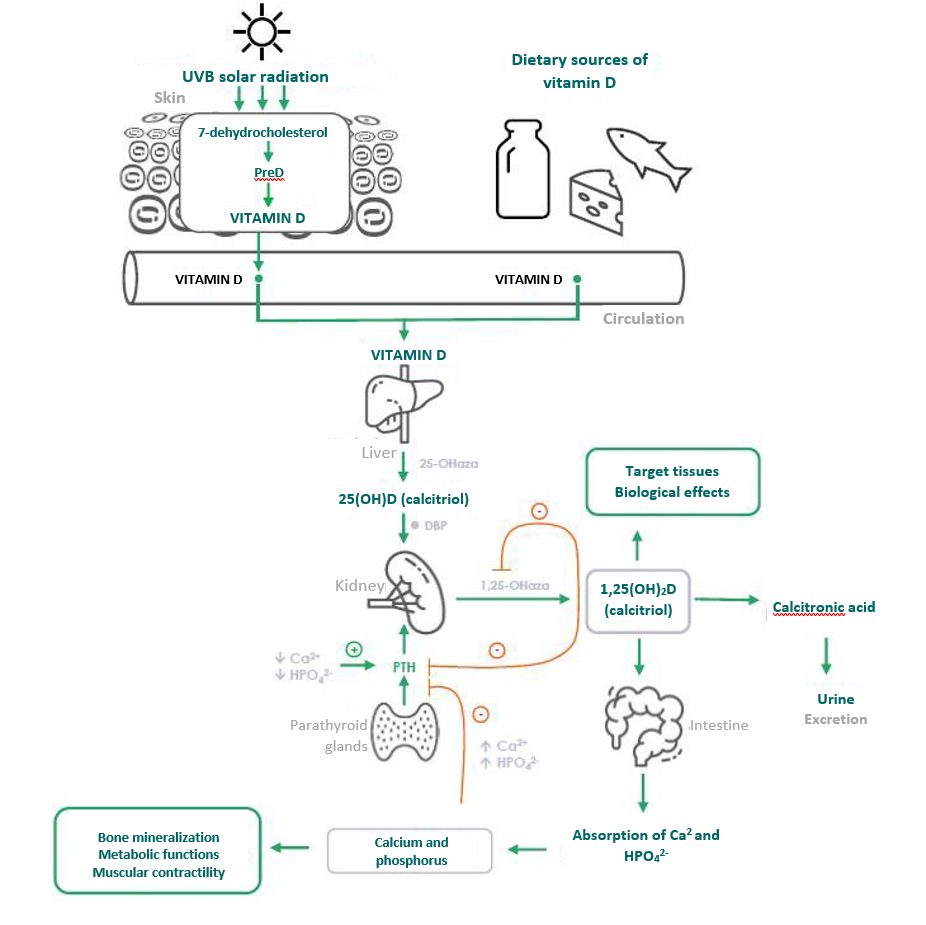
Fig. 1 Vitamin D metabolism and mechanisms regulating vitamin D levels in the body. Own graphic based on Holick 2006 [6].
The intrinsic regulation of the concentration of the active vitamin D metabolite 1,25(OH)2D makes it difficult to overdose.Safety
Vitamin D is considered generally safe, as its levels in the body are internally regulated and maintained within the range of physiological concentrations. Vitamin D supplementation by patients with vitamin D deficiency is generally very well tolerated and toxic levels are rarely observed in clinical practice [7]. According to the latest vitamin D dosage standards developed by the EFSA (European Food Safety Authority) and the IOM (Institute of Medicine) in the USA, the safe daily dose for adults is 4,000 IU/day, which can be increased to 10,000 IU in cases of malabsorption or pathological obesity (after medical consultation) [8]. According to the recommendations of the GIS in Poland, vitamin D in a dose of 4,000 IU is available as a dietary supplement (for healthy persons over 75 years of age) [9], while higher doses are available as medicinal products. The upper safety limit is based on the determination of the correlation between serum 25(OH)D levels and the risk of hypercalcaemia (excessive calcium levels in the blood) [10-13]. A dose of 95 µg/day (3800 IU/day) used chronically has been shown to be the lowest dose that can increase the risk of hypercalcaemia in healthy adults; however, elevated serum calcium levels were found in only six people consuming 95 µg/day for 3 months [14]. Another study compared the safety of 5-month vitamin D supplementation at doses of 100 µg/day (4000 IU) and 25 µg/day (1000 IU). Maximum serum 25(OH)D levels were 48 ng/mL (120 nmol/L) and 40 ng/mL (100 nmol/L), respectively, and neither serum calcium levels nor the calcium/creatinine ratio in the stool changed significantly during the study [12]. A recent study involving 373 older healthy subjects with vitamin D deficiency who were given 400, 4,000 and 10,000 IU daily for 3 years showed that hypercalcaemia (total serum calcium > 2.55 mmol/L) occurred in only 0.3 and 9% (in the 400, 4,000 and 10,000 IU/day groups, respectively), while hypercalciuria (24-hour urinary calcium excretion above 7.5 mmol/day) was detected in 17, 22 and 31% (in the corresponding groups) [15].Toxic doses
Due to the broad therapeutic index (the ratio of the drug dose causing intoxication symptoms to the therapeutically effective dose), vitamin D overdose is rare and only occurs with excessive doses. Taking 30 000 IU of vitamin D daily for more than 3 months is considered a toxic dose. With such supplementation, according to an IOM report published on 30 November 2010, serum 25(OH)D concentrations can reach 200 ng/mL, which carries a high risk of hypercalcaemia and calcium metabolism disorders [16].Causes for overdose
Vitamin D overdose leading to overt hypercalcaemia is rare and may result from endogenous overproduction of the active metabolite 1,25(OH)2D via 1-α-hydroxylation in abnormal macrophages, as is encountered in sarcoidosis (a chronic interstitial lung disease, less commonly involving other organs), or through the release of vitamin D from adipose tissue stores when fat mass is lost rapidly. Excess vitamin D is associated with increased intestinal absorption of calcium and bone resorption (absorption of minerals including calcium by bones) [17]. In the scientific literature, cases of vitamin D overdose after very high doses have also been reported; these are rarely caused by errors related to the manufacture of the preparations, more often there is a mistaken (incorrect dosing) or deliberate (following information from unverified sources) intake of vitamin D preparations at extremely high doses. high doses are particularly dangerous for infants and young children, due to poorly developed self-regulatory mechanisms. The scientific literature describes cases of infants who took vitamin D doses up to 25 times higher than recommended, over a period of several months, resulting in hypervitaminosis D, severe hypercalcaemia manifested by significant dehydration, vomiting, constipation, lethargy and weight loss [18-19]. Rocha and Santos described the case of a 19-year-old man who used a parenteral (intravenously administered) preparation of vitamins A, D and E for veterinary use (5 000 000 IU vitamin D in a 100 ml vial) to achieve increased muscle mass gain. Over the course of a year, he took 300 mL of the preparation (15 000 000 IU/year), resulting in weight loss, nausea and vomiting, as well as hypervitaminosis (serum 25(OH)D concentration was 150 ng/mL) and hypercalcaemia [20]. Sometimes physicians choose to treat patients with high doses of vitamin D that exceed standard recommendations. Cases have been described of patients who were prescribed high doses of vitamin D for various ailments. It was found that patients were taking 2,220,000 – 60,000,000 IU of vitamin D over a period of 4-7 weeks for various conditions. The use of megadoses resulted in hypervitaminosis (25(OH)D in the range of 164-1161 ng/mL) and hypercalcaemia (serum calcium levels in the range of 11.0-15.7 mg/dL), which was clinically manifested by excessive lethargy, nausea and vomiting, polyuria and renal dysfunction [21-24].Summary
- Vitamin D used in doses consistent with recommendations is safe.
- Vitamin D overdose is very rare, due to the fact that vitamin D is a physiological substance, its adequate levels are regulated by the body.
- Cases of vitamin D overdose have been reported in the literature due to administration of very high doses (>30 000/day) over a long period of time.
- Overdose causes hypervitaminosis D, hypercalcaemia, which lead to abdominal pain, vomiting, dehydration, lack of appetite, fatigue and polyuria. No case of overdose has resulted in the death of the patient.
References
- Christakos S, Li S, De La Cruz J, Bikle DD. New developments in our understanding of vitamin metabolism, action and treatment. Metabolism. 2019 Sep;98:112-120. Doi: 10.1016/j.metabol.2019.06.010. Epub 2019 Jun 19. PMID: 31226354; PMCID: PMC6814307.
- Rusińska A, Płudowski P, Walczak M, Borszewska-Kornacka MK, Bossowski A, Chlebna-Sokół D, Czech-Kowalska J, Dobrzańska A, Franek E, Helwich E, Jackowska T, Kalin M, Konstantynowicz J, Książyk J, Lewiński A, Łukaszkiewicz J, Marcinowska-Suchowierska E, Mazur A, Michałus I, Peregud-Pogorzelski J, Romanowska H, Ruchała M, Socha P, Szalecki M, Wielgoś M, Zwolińska D, Zygmunt A. Zasady suplementacji i leczenia witaminą D – nowelizacja 2018 r. Standardy Medyczne/Pediatria. 2018;15:531-559.
- Silva MC, Furlanetto TW. Intestinal absorption of vitamin D: a systematic review. Nutr Rev 2018; 76: 60–76
- Jones G. Pharmacokinetics of vitamin D toxicity. Am J Clin Nutr. 2008 Aug;88(2):582S-586S.
- Norman AW: Sunlight, season, skin pigmentation, vitamin D, and 25-hydroxyvitamin D: Integral components of the vitamin D endocrine system. Am J Clin Nutr 1998; 67: 1108–1110.
- Holick MF. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc. 2006 Mar;81(3):353-73. Doi: 10.4065/81.3.353. PMID: 16529140.
- Nowson CA. Prevention of fractures in older people with calcium and vitamin D. Nutrients. 2010;2:975-84.
- Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK, Durazo-Arvizu RA, Gallagher JC, Gallo RL, Jones G, Kovacs CS, Mayne ST, Rosen CJ, Shapses SA. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96:53-58.
- Uchwała GIS nr 1/2021 Zespołu ds. Suplementów Diet zmieniająca Uchwałę nr 4/2019 w sprawie wyrażania opinii dotyczącej maksymalnej dawki witaminy D w zalecanej dziennej porcji w suplementach diety.
- Gallagher JC, Sai A, Templin T 2nd, Smith L. Dose response to vitamin D supplementation in postmenopausal women: a randomized trial. Ann Intern Med. 2012 Mar 20;156(6):425-37. Erratum in: Ann Intern Med. 2012 May 1;156(9):672.
- Vieth R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr. 1999 May;69(5):842-56.
- Vieth R, Chan PC, MacFarlane GD. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am J Clin Nutr. 2001 Feb;73(2):288-94.
- Bischoff-Ferrari HA, Shao A, Dawson-Hughes B, Hathcock J, Giovannucci E, Willett WC. Benefit-risk assessment of vitamin D supplementation. Osteoporos Int. 2010 Jul;21(7):1121-32.
- Narang NK, Gupta RC, Jain MK. Role of vitamin D in pulmonary tuberculosis. J Assoc Physicians India. 1984 Feb;32(2):185- 8.
- Billington EO, Burt LA, Rose MS, Davison EM, Gaudet S, Kan M, Boyd SK, Hanley DA. Safety of High-Dose Vitamin D Supplementation: Secondary Analysis of a Randomized Controlled Trial. J Clin Endocrinol Metab. 2020 Apr 1;105(4):dgz212. Erratum in: J Clin Endocrinol Metab. 2021 Mar 25;106(4):e1932.
- Jones G. Pharmacokinetics of vitamin D toxicity. Am J Clin Nutr. 2008 Aug;88(2):582S-586S. doi: 10.1093/ajcn/88.2.582S. PMID: 18689406.
- Rizzoli R, Stoermann C, Ammann P, Bonjour JP. Hypercalcemia and hyperosteolysis in vitamin D intoxication: effects of clodronate therapy. Bone. 1994 Mar-Apr;15(2):193-8.
- Ketha H, Wadams H, Lteif A, Singh RJ. Iatrogenic vitamin D toxicity in an infant–a case report and review of literature. J Steroid Biochem Mol Biol. 2015 Apr;148:14-8.
- Bilbao NA. Vitamin D Toxicity in Young Breastfed Infants: Report of 2 Cases. Glob Pediatr Health. 2017 Sep 19;4:2333794X17731695.
- Rocha PN, Santos CS, Avila MO, Neves CL, Bahiense-Oliveira M. Hypercalcemia and acute kidney injury caused by abuse of a parenteral veterinary compound containing vitamins A, D, and E. J Bras Nefrol. 2011 Dec;33(4):467-71
- Kaur P, Mishra SK, Mithal A. Vitamin D toxicity resulting from overzealous correction of vitamin D deficiency. Clin Endocrinol (Oxf). 2015 Sep;83(3):327-31.
- Koul PA, Ahmad SH, Ahmad F, Jan RA, Shah SU, Khan UH. Vitamin d toxicity in adults: a case series from an area with endemic hypovitaminosis d. Oman Med J. 2011 May;26(3):201-4.
- Pandita KK, Razdan S, Kudyar RP, Beigh A, Kuchay S, Banday T. “Excess gooD can be Dangerous”. A case series of iatrogenic symptomatic hypercalcemia due to hypervitaminosis D. Clin Cases Miner Bone Metab. 2012 May;9(2):118-20.
- Chowdry AM, Azad H, Najar MS, Mir I. Acute kidney injury due to overcorrection of hypovitaminosis D: A tertiary center experience in the Kashmir Valley of India. Saudi J Kidney Dis Transpl. 2017 Nov-Dec;28(6):1321-1329.
Biographical note

tel.: +48 61 625 27 00
fax: +48 61 625 27 01
office@chc.com.pl
bd@chc.com.pl
purchasing@chc.com.pl
rd@chc.com.pl
BDO: 000102401
Wysogotowo
ul. Batorowska 52
62-081 Przeźmierowo
POLAND
District Court for Poznań - Nowe Miasto and Wilda in Poznań, the 8th Commercial Division of the National Court
KRS 0000871229
NIP 781-00-41-371
Share capital: 36 100 000PLN
© Curtis Health Caps. All rights reserved.


